Use the Build Study page to create or edit the Study Details, CRFs, Event Definitions, Subject Group Classes, Rules, Sites, and Users. Follow this basic workflow to design or edit a Study:
- The Study must exist, having been created using the Administration module.
- Change the current Study to the Study you want to work on. You must work at the Study level; the Build Study features are not available at the Site level.
- Access the Build Study page by selecting Tasks > Build Study.
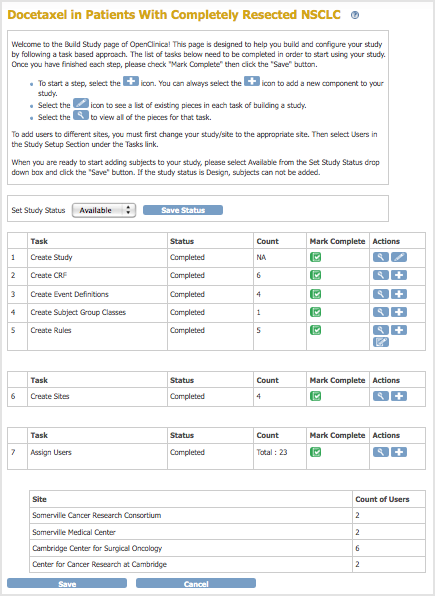
The Build Study page opens for the current Study, presenting a table of the main Tasks, with status information for each, and options to view details, create new elements, and edit the existing configuration. - Set the Study Status, if needed:
- When you are creating the Study, set the Status to Design, then click the Save Status button.
- After you’ve created the Study and are making edits, set the Status to Available, then click the Save Status button.
For more information, see Status of Study.
- Add to or edit the Study configuration by clicking the appropriate icon for Tasks 1 to 5 in the table. For details, see the following sections:
- Create Study: Edit Details
- Create and Modify Case Report Forms (CRFs)
- Create and Modify Event Definitions
- Create and Modify Subject Group Classes
- Create and Modify Rules
When you begin to work on a Task, the Task’s Status changes from Not Started to In Progress.
- When you have finished a Task, select the checkbox in the Mark Complete column, then click the Save button. The Task’s Status changes from In Progress to Complete. Note that you can make changes to a Task, even when its Status is Complete.
- After Tasks 1 to 5 are marked complete, add or modify Sites.
- Add or modify Users.
- If you haven’t already done so, set the Study Status to Available, and click the Save Status button.
- After the Status is Available, add Subjects to the Study and start collecting Study data.
Build Study Page After Completing Study Setup:
5.2.1 Status of Study
Some actions update the Study status automatically, while some Status values are set by a user with appropriate permissions using the Set Study Status drop-down list on the Build Study page. These Status values also apply to Sites.
Status | Use to | Comments |
Design (also displayed in OpenClinica as Pending) | Build a Study | - When you create a Study, the status is automatically set to Design.
- Subjects cannot be enrolled in a Study if the status is Design (the Add Subject button is unavailable).
- Once all the Build Study tasks are complete, for data entry to begin, the status must be changed to Available.
|
Available | Provide access to the Study to users (for Subject enrollment, data entry, editing, etc.) as the last step of building a Study. | - Subjects cannot be enrolled in a Study until the status is set to Available.
|
Frozen | Prevent Site users from signing, entering, and editing data, but allow Study level users to edit data as needed to close Notes and Discrepancies. | - Site users can view data but cannot enter or edit data.
- Investigators cannot sign Events or Case Books.
- Study-level users can view data and can close Discrepancies. Closing Discrepancies may involve editing data, which they can do via Notes and Discrepancies if they click the icon to View within record for CRFs having a status of Completed. It should be noted that this also allows users to change values for other items on the record that may not have an associated discrepancy. Your internal procedures should explicitly state what can/cannot be edited.
- As always, all data changes are tracked in the Audit Log.
|
Locked | Prevent all users from entering or editing data. | - Neither Site nor Study level users can sign CRFs, enter or edit data, or create or edit Notes and Discrepancies.
- Viewing, printing, and extracting data are permitted.
|
Removed | Make the study inaccessible to end users. | - This status is set automatically when a Study is Removed.
- Only Administrators can remove a Study.
|

